My nurses do a great job keeping the clinical service running, especially handling prescriptions and the nonsense sometimes required by insurers. Treating children with chronic kidney disease means prescribing a lot of medications not approved specifically for use in children. Sometimes we must jump through flaming hoops to get the job done.
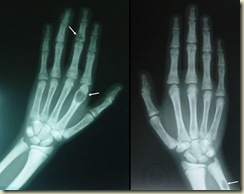
We prescribed calcium acetate, a medication used to bind phosphate. As the kidneys lose function, they cannot eliminate phosphate from the body. Dietary reduction helps, but since most organisms store energy in phosphate molecules like ATP, it cannot be eliminated entirely. Patients can take calcium or other medications with meals to bind to the phosphate in the gut. Calcium phosphate cannot be absorbed into the body. Instead, it is fecally excreted (or pooped out, as I tell my patients). Excess phosphate (along with lack of the active form of vitamin D) causes bone problems in many patients with chronic kidney disease, including secondary hyperparathyroidism (shown in x-ray).
Being generally cognizant of insurance limitations, we generate most prescriptions for the generic drug, in this case:
Calcium Acetate 667 mg three times daily with meals
Today, we received a fax:
Patient must fail a 2-week trial of PhosLo before calcium acetate can be prescribed.
Yup, same stuff. So we wrote back and gave permission to prescribe PhosLo. Duh!
Thank you, Nebraska Medicaid, for providing today’s blog fodder. And a lot of head-shaking in our office!





No comments:
Post a Comment